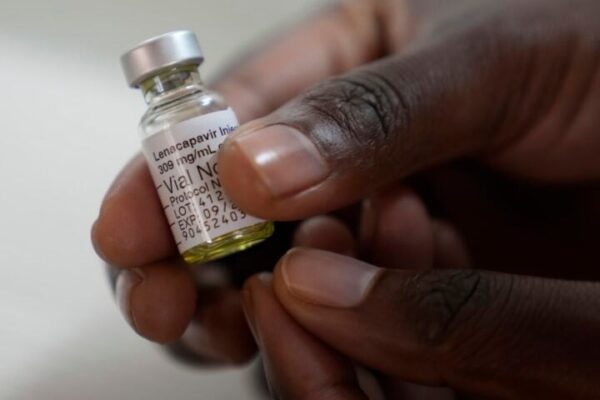
The National Agency for the Control of Aids (NACA) has announced that commodities for the rollout of Lenacapavir as a new HIV prevention option are expected in Nigeria by March 2026.
In a statement released by the head of public relations, Toyin Aderibigbe, the agency revealed that preparations for a phased introduction are already underway, with readiness assessments completed in ten states, including Lagos, Kano, Benue, and the Federal Capital Territory.
Lenacapavir is an injectable treatment and a convenient alternative to daily oral prevention drugs.
The agency says the injectable pre-exposure prophylaxis, taken just twice yearly, is part of efforts to strengthen HIV prevention and accelerate progress toward epidemic control.
Read Also
The drug is expected to be available in Nigeria and 119 other low- and middle-income countries at an affordable price of $40 per person annually, thanks to voluntary licensing agreements with generic manufacturers.
Regulatory approval has been secured from the National Agency for Food and Drug Administration and Control, while a national training of trainers has been conducted in Abuja, followed by step-down training for healthcare workers in implementing states.
NACA says information, education, and communication materials have also been developed to support community engagement, as Nigeria moves toward a structured and quality-assured rollout of the new prevention option.